Categorisation of refrigerant substances and mixtures
Cooling systems require a fluid passing through the system to transfer heat from the object to be cooled through the heat exchanger itself. In a direct cooling system, a vaporising refrigerant substance passes through the cooling mechanism. When the substance is a separate liquid, or so-called refrigerant mixture (non-vaporising), this is referred to as an indirect cooling system.
Refrigerant substances
Refrigerants are generally hydrocarbons whose hydrogen atoms have been replaced by halogen molecules through processing. In legislation, refrigerants are categorised precisely on the basis of halogen molecules. Halogenated hydrocarbons can be divided into the following groups based on their halogen molecules:
CFC refrigerants are fully halogenated hydrocarbons containing chlorine, fluorine and carbon but no hydrogen (Chloro-Fluoro-Carbon). They are refrigerants with a high ozone and significant greenhouse effect.
HCFC refrigerants are partially halogenated hydrocarbons containing chlorine, fluorine, carbon and hydrogen (Hydro-Chloro-Fluoro-Carbon). They have a negligible effect on the ozone layer but a significant adverse effect on greenhouse gas emissions.
HFC refrigerants in turn are partially halogenated hydrocarbons containing fluorine, carbon and hydrogen (Hydro-Fluoro-Carbon). Like HCFCs, they are ozone-free but have a significant greenhouse effect.
PFC refrigerants are fully halogenated hydrocarbons containing only fluorine and carbon (Per-Fluoro-Carbon). Like the above, PFCs are ozone-free but have a significant greenhouse effect.
HFO refrigerants are partially halogenated hydrocarbons containing fluorine, carbon and hydrogen (Hydro-Fluoro-Olefin). Compared to the above, HFO refrigerants are both ozone-free and have low greenhouse effects.
HFC and PFC refrigerants are so-called hydrofluorocarbons and are commonly referred to as F-gases. HFO refrigerants are also substances classified as F-gases. HFO substances differ from HFCs in molecular structure: HFO substances have a so-called double bond between two carbon atoms; HFCs have only single bonds between carbon atoms.
The F-Gas Regulation treats HFO/HFC mixtures like pure HFCs but requires only reporting for pure HFOs.
In addition to the above groups, there is a group of refrigerants that do not contain any halogen molecules. These refrigerants are referred to as so-called natural refrigerants and include, for example, pure hydrocarbons, ammonia and carbon dioxide. These substances occur naturally in nature, are not harmful to the ozone layer in the atmosphere and have a zero or near zero greenhouse effect. Natural refrigerants can be divided into two groups:
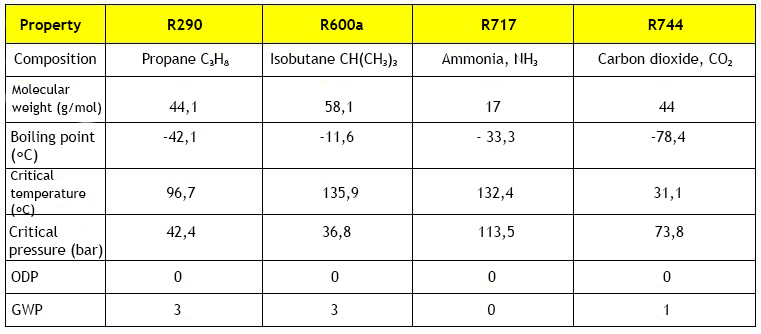
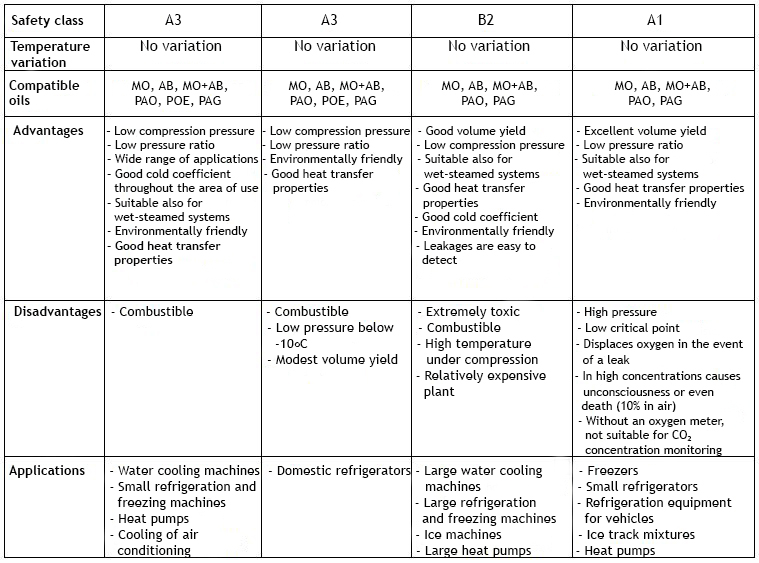
HC refrigerants are pure hydrocarbons (Hydro-Carbon) such as propane (R290) and butane (R600) (propylene, isobutane and propylene) that are ozone-free refrigerants with a practically negligible GWP (Global Warming Potential). However, they are highly flammable, which limits their usability.
Inorganic Compounds include ammonia (R717) and carbon dioxide (R744), which are the most commonly used. Both of the above substances are harmless to the ozone layer; ammonia has a GWP of 0 and carbon dioxide of 1 when the corresponding values for CFCs and HFCs are from 1300 upwards. Other inorganic compounds include water and air. The downside of ammonia is its toxicity, as well as its concentration and temperature dependent explosive properties in confined spaces. Otherwise ammonia is not easily flammable. Because of these properties, the use of ammonia requires special machine room space. Carbon dioxide in turn displaces oxygen at high concentrations, which can cause various symptoms and even suffocation.
Labelling of refrigerants
In refrigeration technology, refrigerants are designated by the internationally agreed symbol R (refrigerant) followed by numerals. For halocarbons (CFC, HCFC, HFC) the number indicates the number of carbon, hydrogen and fluorine atoms, respectively. If there is more than one carbon atom in the molecule, the positions of the atoms may vary while the composition remains unchanged. These so-called monomers are distinguished by a letter following the numerical code (e.g. R134a). Some so-called azeotropic halogenated hydrocarbon mixtures are numbered according to their invention sequence from R500 upwards and for zeotropic refrigerant mixtures starting with R400. The numbers from 600 upwards are reserved for butanes. Inorganic compounds have an agreed labelling policy starting with the symbol R700. The number in excess of 700 indicates the molecular weight of the substance, for example ammonia is R717 as the molecular weight of ammonia is 17.
Refrigerant mixtures
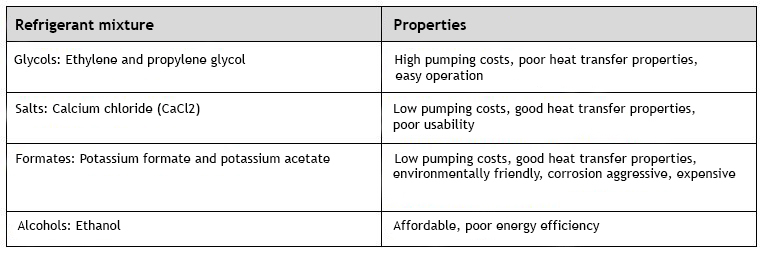
Refrigerant mixtures are used for indirect cooling and indirect condensation. The most common aqueous solutions are ethylene glycol water, propylene glycol water, ethanol water, calcium chloride (CaCl2) water, potassium formate water and ammonia water. The properties of the different refrigerant mixtures differ in their environmental, material, health, pumpability and heat transfer properties.
Aqueous solutions of non-freezing liquids that either occur in nature or, when released into the environment, decompose into compounds that are harmless to the environment are primarily used as refrigerant mixtures. The temperature of refrigerant mixtures varies depending on the application; for example, the design temperatures may be -12/-9 ° C and the operating temperatures -10/-8 ° C.
Indirect cooling systems have grown in popularity due to the environmental problems caused by CFCs and HCFCs, changes in legislation and the safety risks related to ammonia refills. These systems have been installed mainly in the food industry and at ice rinks, but they are also suitable for cold storage in retail stores.
Although the amount of refrigerant mixture required for an indirect cooling system is often very large, the refrigerant fillings are quite small. The choice of refrigerant mixture and the resulting design, construction and maintenance of the system have a big impact on the system’s investment costs, efficiency and usability.
Refrigerant mixture choices and their properties
The most important aspect in designing an indirect cooling system is selecting a refrigerant mixture with a good heat transfer capacity. Thanks to its excellent heat transfer capacity and many other positive properties, water is an excellent heat transfer fluid for indirect cooling systems. However, in applications where the temperature drops below zero, the use of a freezing point lowering agent is required. The choice of the agent should take into account factors such as the thermal conductivity of the liquid, the freezing point, its compatibility with metals, alloys and seals, its stability in use, non-toxicity, biodegradability, a high flash point and a reasonable cost.
The properties of different refrigerant mixtures differ in their environmental, material, health, pumpability and heat transfer properties. In terms of the thermodynamic properties of refrigerant mixtures, viscosity has the greatest effect on the cooling power of the refrigeration system and the required heat transfer surface area. The heat transfer surface area in turn has an impact on investment costs. The most commonly used refrigerant mixtures include glycols (propylene and ethylene), alcohols (ethanol), and both organic (potassium formate, potassium acetate) and inorganic salts (calcium chloride).
Water-alcohol / glycol mixtures
Monohydric alcohols, such as methanol and ethanol, are effective in lowering the freezing point and are also better than inorganic salts for use with most common building materials. However, the toxicity of methanol makes it unsuitable for food industry applications. The biggest weakness of ethanol is its low boiling point, which at the same time lowers the boiling point of ethanol mixtures.
Ethylene and propylene glycol lower the freezing point like alcohols, but at the same time they raise the boiling point of the mixture. Due to its ease of use, food-grade propylene glycol is the most commonly used refrigerant for indirect cooling in the food industry. Its strengths are its high viscosity and non-toxicity, while its downsides are the higher price and lower heat transfer capacity.
Organic salts
Organic salts, such as potassium formate, provide a solution when looking for an effective heat transfer fluid for applications that must withstand -20oC or colder conditions and be compatible with different materials. The strengths of organic salts are their good heat transfer capacity, high specific heat capacity and non-toxicity. Their disadvantage, in turn, is their higher corrosivity, which must be taken into account when choosing materials. Corrosion inhibitors can be used to reduce the corrosion of aluminium in organic salt mixtures as needed. Organic salt mixtures themselves have pH buffering properties, and their degradation products are not corrosive, which are benefits.
Inorganic salts
Conventional salt mixtures containing potassium carbonate or calcium chloride offer good thermal conductivity. They are non-toxic, inexpensive and effective at lowering the freezing point of the liquid. However, the main problem with chloride containing solutions is the pitting corrosion of metals. Only titanium is completely immune to pitting corrosion. This reduces the usability of the solution in simple industrial refrigeration systems. With the right inhibitor, it is possible to use calcium chloride with copper, titanium, carbon steel and cast iron.
In order to select the most suitable fluid for your cooling system, the first step is to determine whether it is a direct or indirect system. The importance of environmental, material, health, pumpability and heat transfer properties for the intended use should then be assessed. Algol Chemicals offers a product portfolio that includes solutions for both direct and indirect cooling systems. Through the experience and know-how we have gained together with our suppliers and clients, we can help you choose the best coolant option.